Page 23 - ISAKOS 2021 Newsletter Volume 1
P. 23
CURRENT CONCEPTS
Preclinical results on the use of iPS cells have shown promise for their possible future use.
Growth Factors
Growth factors initiate and control a large number of cellular mechanisms; as such, they are used in tissue engineering to encourage superior chondrogenesis and cell proliferation. Insulin-like growth factor, fibroblast growth factor, and the transforming growth factor-beta (TGF- ) superfamily, which consists of bone morphogenic proteins (BMP-2, 4, 6, 7), cartilage-derived morphogenic proteins (CDMP-1, 2), and transforming growth factor beta-1 (TGF- ) are mainly involved in cartilage regeneration. These factors (along with other, less-notable factors) play vital roles in promoting cell proliferation and proteoglycan synthesis and in inhibiting catabolic processes.
Because of their reparative and regenerative functions, growth factors can be used to initiate better chondral repair. Platelet-rich plasma, autologous conditioned plasma, and bone marrow concentrate are considered to be abundant in growth factors and have been used in clinical practice. These are manufactured by the concentration of blood components or bone marrow aspirate by means of a centrifugation process or a system that concentrates the native growth factors that are present within the sample. There is still uncertainty regarding whether such blood / marrow-derived products promote chondrogenic differentiation of MSCs. Although these therapies are theoretically an easily available source of growth factors, a major pitfall is that autologous therapies lack standardization.
Scaffolds
Chondral Layer
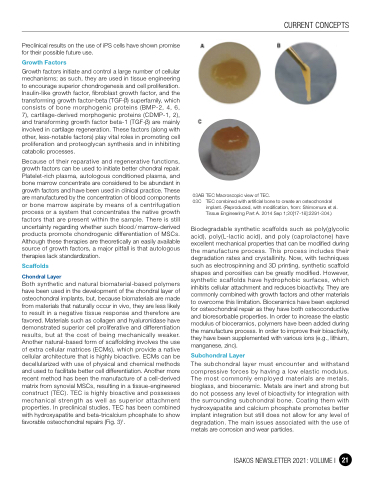
Both synthetic and natural biomaterial-based polymers have been used in the development of the chondral layer of osteochondral implants, but, because biomaterials are made from materials that naturally occur in vivo, they are less likely to result in a negative tissue response and therefore are favored. Materials such as collagen and hyaluronidase have demonstrated superior cell proliferative and differentiation results, but at the cost of being mechanically weaker. Another natural-based form of scaffolding involves the use of extra cellular matrices (ECMs), which provide a native cellular architecture that is highly bioactive. ECMs can be decellularized with use of physical and chemical methods and used to facilitate better cell differentiation. Another more recent method has been the manufacture of a cell-derived matrix from synovial MSCs, resulting in a tissue-engineered construct (TEC). TEC is highly bioactive and possesses mechanical strength as well as superior attachment properties. In preclinical studies, TEC has been combined with hydroxyapatite and beta-tricalcium phosphate to show favorable osteochondral repairs (Fig. 3)1.
03AB TEC Macroscopic view of TEC.
03C TEC combined with artificial bone to create an osteochondral
implant. (Reproduced, with modification, from: Shimomura et al. Tissue Engineering Part A. 2014 Sep 1;20[17-18]:2291-304.)
Biodegradable synthetic scaffolds such as poly(glycolic acid), poly(L-lactic acid), and poly (caprolactone) have excellent mechanical properties that can be modified during the manufacture process. This process includes their degradation rates and crystallinity. Now, with techniques such as electrospinning and 3D printing, synthetic scaffold shapes and porosities can be greatly modified. However, synthetic scaffolds have hydrophobic surfaces, which inhibits cellular attachment and reduces bioactivity. They are commonly combined with growth factors and other materials to overcome this limitation. Bioceramics have been explored for osteochondral repair as they have both osteoconductive and bioresorbable properties. In order to increase the elastic modulus of bioceramics, polymers have been added during the manufacture process. In order to improve their bioactivity, they have been supplemented with various ions (e.g., lithium, manganese, zinc).
Subchondral Layer
The subchondral layer must encounter and withstand compressive forces by having a low elastic modulus. The most commonly employed materials are metals, bioglass, and bioceramic. Metals are inert and strong but do not possess any level of bioactivity for integration with the surrounding subchondral bone. Coating them with hydroxyapatite and calcium phosphate promotes better implant integration but still does not allow for any level of degradation. The main issues associated with the use of metals are corrosion and wear particles.
ISAKOS NEWSLETTER 2021: VOLUME I 21